 
This is how the redox equations are balanced. Now, the equation is balanced with 2 Chloride’s (Cl) with total charge -2 and 3 Chromium’s with total charge +3 on both sides. Making sure that the total charge is the same on both sides of the equation is known as balancing by charge. By balancing the mass, one may make sure that each element is present in equal amounts on both the product and reactant sides. That's it! the reaction is now properly balanced □. To balance the unbalanced chloride molecule charges, we add 2 in front of the chloride on L.H.S. Mass and charge must be equal in net ionic equations. Mass of Reactants = Mass of Products \small \text+ 19\text O_2 \longrightarrow 12\text C\text O_2 + 14\text H_2\text O 2 C 6 H 14 + 19 O 2 ⟶ 12 C O 2 + 14 H 2 O Find more Chemistry widgets in WolframAlpha. 
With this in mind, when balancing chemical reactions, the goal is to equilibrate the masses of reactants and products of the equation: Get the free 'NET IONIC EQUATION CALCULATOR' widget for your website, blog, Wordpress, Blogger, or iGoogle. 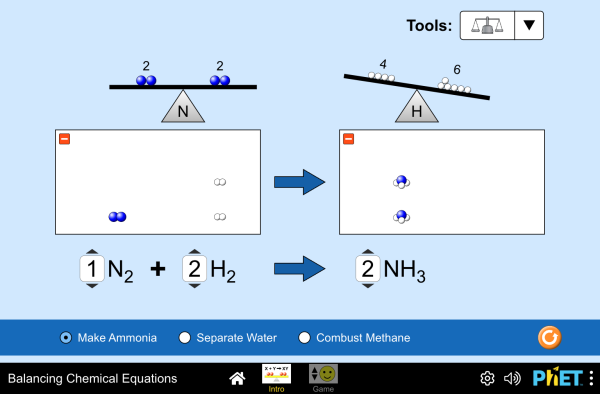
Use uppercase for the first character in the element and lowercase for the second character. The balanced equation will be calculated along with the oxidation states of each element and the oxidizing and reduction agents. Solid potassium chlorate, KClO 3, decomposes to form solid potassium chloride and diatomic oxygen gas. Enter an equation of a redox chemical reaction and press the Balance button. That means that the total mass of reactants and products is the same before and after the reaction. Write a balanced equation describing each of the following chemical reactions. Ionic charges are not yet supported and will be ignored. Step 2: To obtain a balanced equation, now click on 'Balance'. According to Lavoisier's law of conservation of mass, the total mass of a chemical reaction system doesn't change. Instructions To balance a chemical equation, enter an equation of a chemical reaction and press the Balance button. Step 1: First, enter the chemical formula in the text box. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
